
Scale-up
Für das Scale-up von neu erarbeiteten Formulierungen in einer Pilot-Anlage steht im Institut Dr. Schrader Beratungslabor die neueste Generation der bewährten Becomix-Technologie zur Verfügung.

Sie erhalten nicht nur statistische Daten – unsere gerichtsfesten Gutachten belegen zuverlässig die Claims Ihres Produkts.

Absolute Vertraulichkeit ist die Basis der Zusammenarbeit mit unseren Kunden seit mehr als fünf Jahrzehnten.

Qualität, Know-how und Kompetenz in Sachen Kosmetik machen uns zu einem weltweit geschätzten Partner.

Nur bei uns finden Sie fachübergreifendes Know-how in Forschung, Entwicklung, Prüfung und Analytik für den entscheidenden Unterschied im Wettbewerb.
Vom Standard-Prüfdesign bis hin zum individuell angepassten Studien-Design einer Kombinationsstudie, von kleinen Screening-Untersuchungen in der ersten Wirksamkeitsanalyse bis zur großen Studie mit entsprechender statistischer Begleitung der Datenanalyse in der Bio-Statistik.
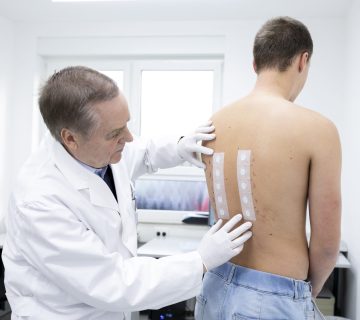
Im Haarbereich werden Anwendungs-Studien aller Art verbunden mit dermatologischer und ophthalmologischer Kontrolle durchgeführt.


Unabhängige galenische Anwendungstechnik für die Kosmetikindustrie ist seit 50 Jahren unsere Aufgabe. Im Rahmen der Galenik erarbeiten wir neue, individuell zugeschnittene Formulierungen für jegliche Art von Kosmetika für Haut-Pflege und Haar-Pflege – speziell auch Sonnenschutz-Produkte sowie Naturkosmetik.
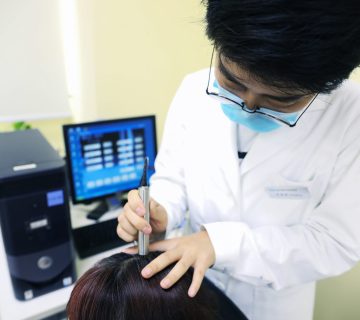
Der analytische Arbeitsbereich steht für chemisch-analytische Untersuchungen von Kosmetika. Der Schwerpunkt der Arbeiten der Abteilung liegt dabei auf der Qualitätssicherung von kosmetischen Produkten.


Der Fachbereich der Forschung ist die Basis vieler Innovationen und Neuerungen, durch die sich das Institut Dr. Schrader immer wieder auszeichnen und empfehlen.
Um den immer größer werdenden Anforderungen des asiatischen Marktes Rechnung zu tragen, wurde 2004 die Institute Dr. Schrader International GmbH gegründet.
Institut Dr. Schrader – Keimzelle weltweit angewandter Verfahren.
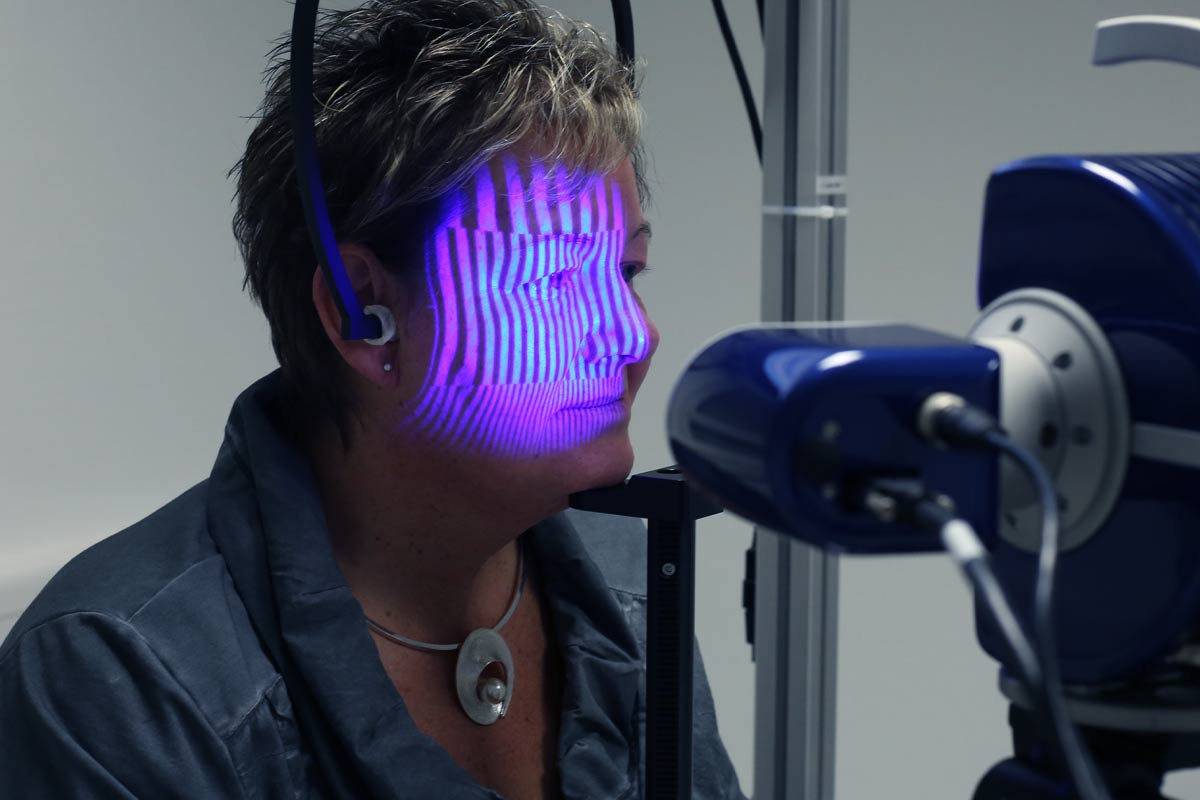
Neue Verfahren als Grundlage für Wirksamkeit und Verträglichkeit kosmetischer Produkte. Fünf Jahrzehnte Ideen und Konzepte für haut- und haarphysiologische Messprinzipien, die heute zu weltweiten Standard-Methoden gereift sind.
Unsere bekanntesten Entwicklungen:

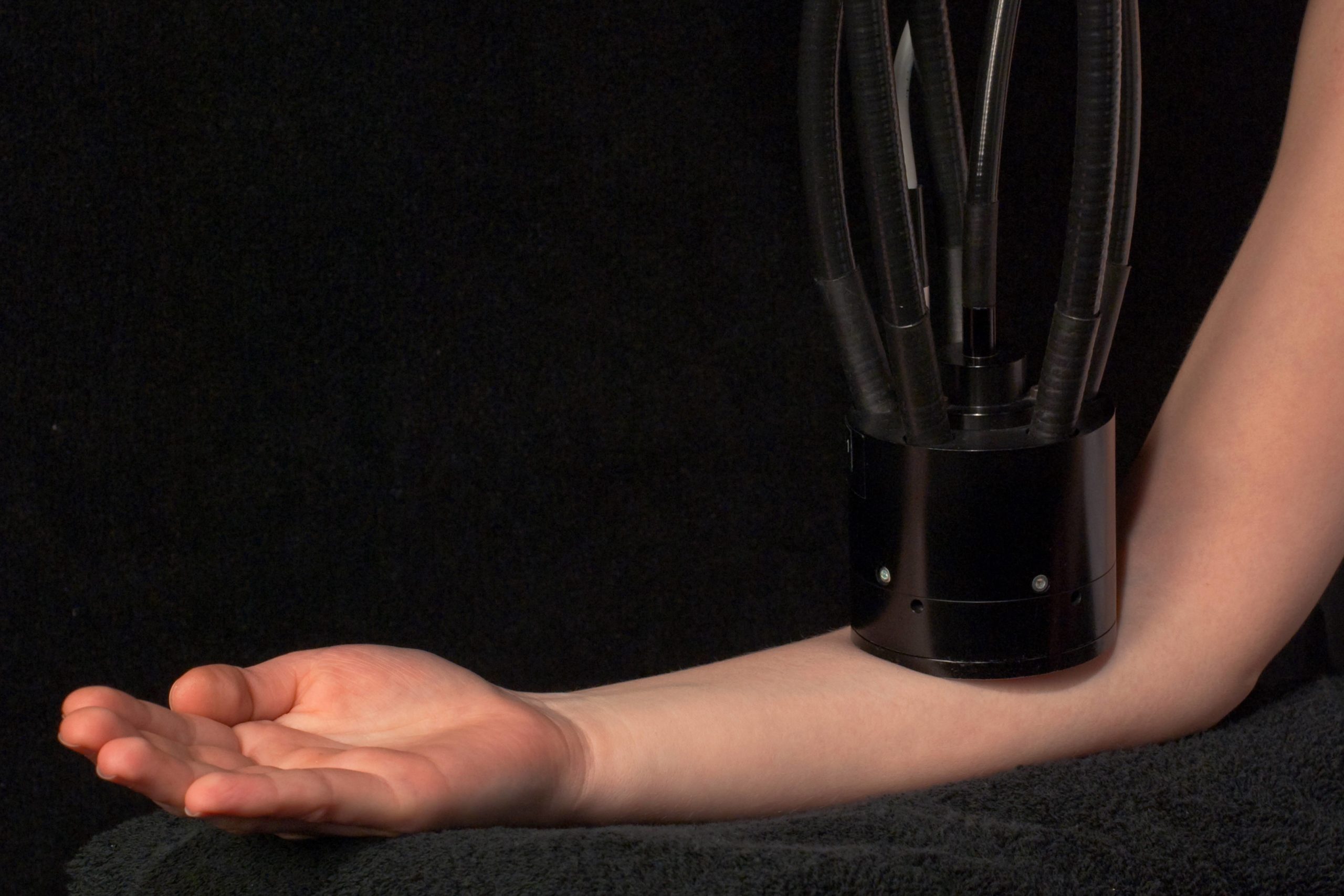


Für das Scale-up von neu erarbeiteten Formulierungen in einer Pilot-Anlage steht im Institut Dr. Schrader Beratungslabor die neueste Generation der bewährten Becomix-Technologie zur Verfügung.

Analyse vor und nach der Behandlung – Es werden Haare mit bekanntem Durchmesser entweder bis zum Haarbruch gedehnt oder nur im elastischen Bereich gedehnt und anschließend entspannt. Mit Hilfe dieser Technik…

Olfaktorische Beurteilung des Körpergeruchs von geübten Bewertern | Sniffing-Test – Die olfaktorische Beurteilung des Körpergeruchs von geübten Bewertern (Sniffern) ist die realistischste Methode, die Wirksamkeit…

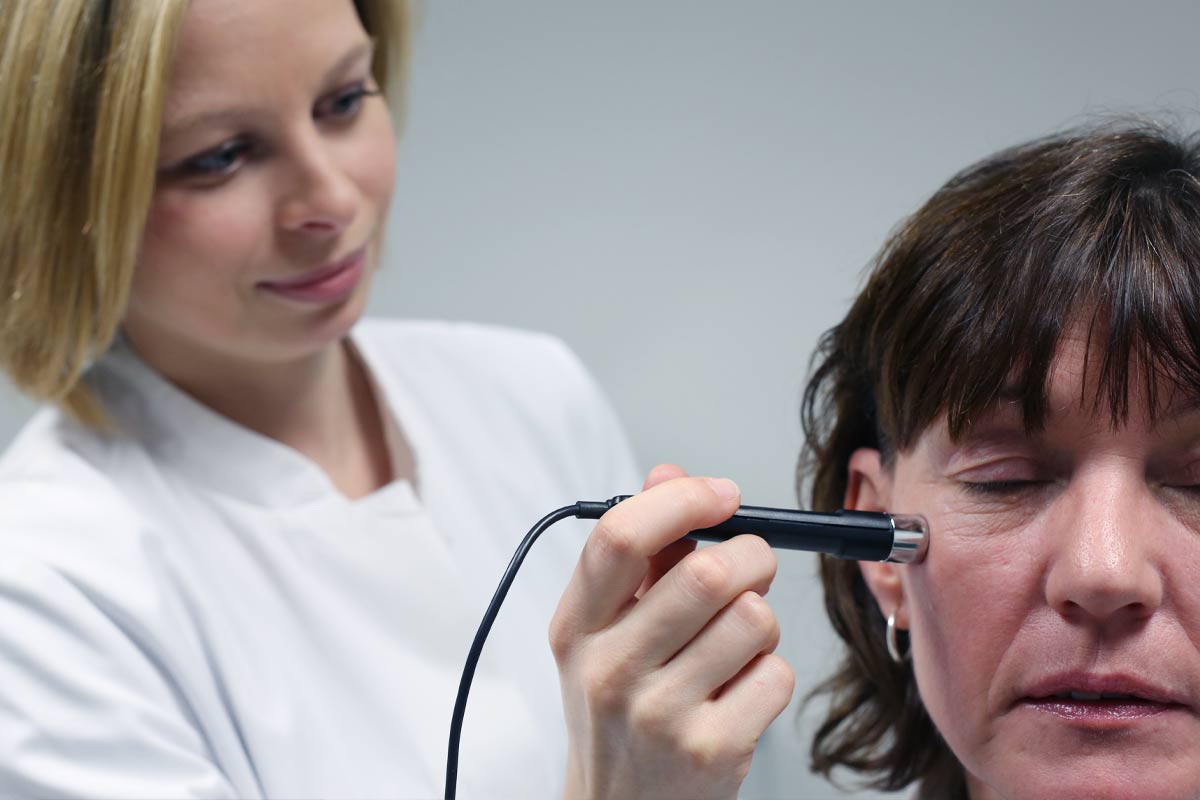
Der Sicherheitsbewertung kosmetischer Mittel kommt eine immer wichtigere Bedeutung zu, auch die gesetzlichen Sicherheitsbestimmungen werden kontinuierlich verändert.

Endlich kommt der Frühling zurück… und damit natürlich auch die Sonne!

Die Institute Dr. Schrader beteiligen sich am 26. Und 27. April 2023

Die Institute Dr. Schrader beteiligen sich am 26. und 27. April 2023 am DGK Fortbildungskurs.

Die neue HDRS-Technik stellt zum ersten Mal einen in-vivo-Ansatz für Sonnenschutzprüfungen …
